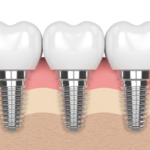
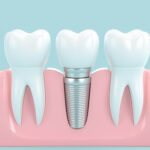
Full loss of teeth is known as total edentulism, and it occurs in a large proportion of the population of adults. Generally, it can occur as after tooth extractions are necessary from either decay, gum disease, or because a patient has not been able to afford the costs associated with dental treatment.
In general, people lose their teeth due to cavities and gum disease. Centers for Disease Control and Prevention figures indicate that by late teenage years, about 7 percent of American report the loss of at least one permanent tooth. Gums are crucial in attaching to teeth at along the gum edge. If plaque is trapped in the gum space, gingivitis (gum infection can occur). As it continues to cover the teeth, it can become harder in the process of becoming tartar. When gingivitis becomes involved in infection under the gum, sometimes gums weaken and soft tissue and bone holding the teeth are damaged. As infection continues, patients can eventually lose teeth or end up at the dentist’s office asking for it to be removed.
Just as in other types of physical ailments, total loss of teeth occurs as a patient grows older. The possibility of edentulism grows steadily over each decade as a patient enters adulthood and increases by even more after the patient turns 70, according to research.